|
This phenomenon is due to the opposite charges on each ion.Ĭovalent bonding is the sharing of electrons between atoms. Most ionic compounds tend to dissociate in polar solvents because they are often polar. 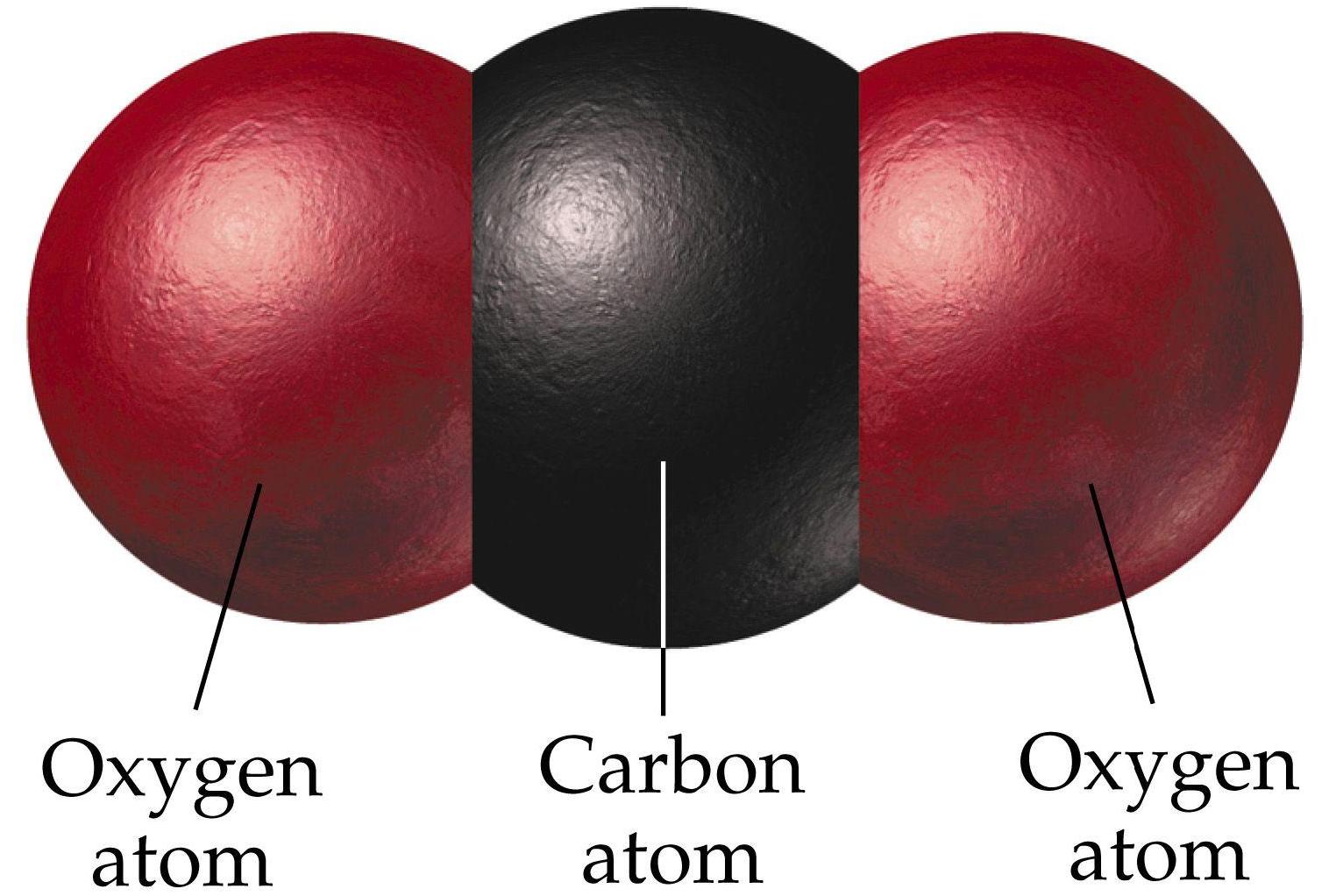
At the ideal interatomic distance, attraction between these particles releases enough energy to facilitate the reaction. However, this reaction is highly favorable because of the electrostatic attraction between the particles. The predicted overall energy of the ionic bonding process, which includes the ionization energy of the metal and electron affinity of the nonmetal, is usually positive, indicating that the reaction is endothermic and unfavorable. This chlorine atom receives one electron to achieve its octet configuration, which creates a negatively charged anion. This creates a positively charged cation due to the loss of electron. 
This sodium molecule donates the lone electron in its valence orbital in order to achieve octet configuration. Carbon dioxide contains covalent bonds only two of them between each oxygen and the centralised carbon atom, for four (4) total per molecule.An ionic bond is a type of chemical bond that involves. In ionic bonds, the net charge of the compound must be zero. Is CO2 ionic or covalent : The covalent compound is formed by two non-metal atoms, in case of CO 2 compound formation, carbon and oxygen are non-metal while the ionic compound is formed by a metal atom and a non-metal atom. The charges on the anion and cation correspond to the number of electrons donated or received. 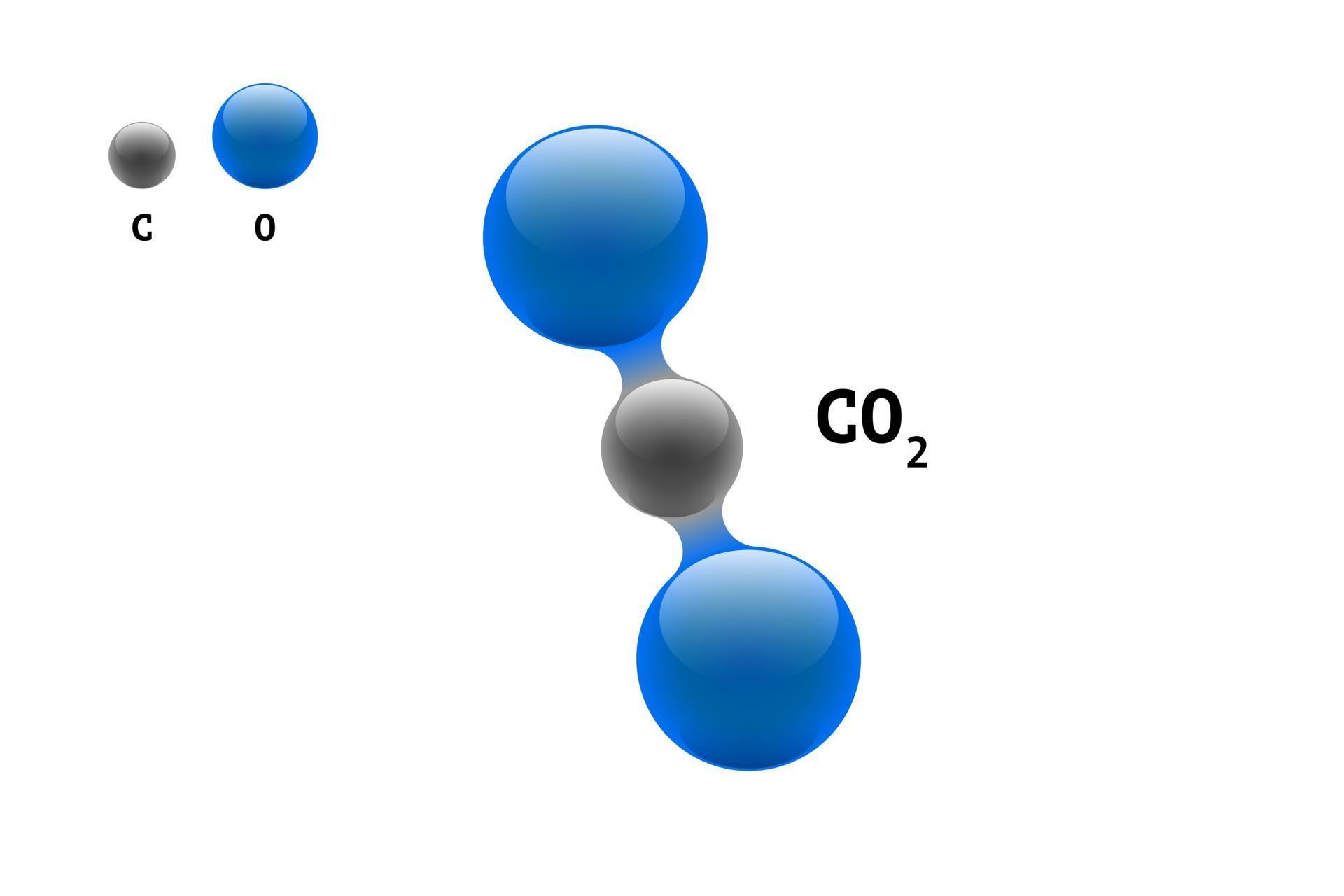
In ionic bonding, more than 1 electron can be donated or received to satisfy the octet rule. Similarly, nonmetals that have close to 8 electrons in their valence shells tend to readily accept electrons to achieve noble gas configuration. Similarly, carbon dioxide, which contains one carbon atom and two oxygen atoms in each molecule, is written as CO2. By losing those electrons, these metals can achieve noble gas configuration and satisfy the octet rule. Ionic bonding is observed because metals have few electrons in their outer-most orbitals. Ionic bonds require an electron donor, often a metal, and an electron acceptor, a nonmetal. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. It is a type of chemical bond that generates two oppositely charged ions. Ionic bonding is the complete transfer of valence electron(s) between atoms. The equilibrium is reached when the number of molecules escaping from the liquid phase is the same as the number of molecules entering it.\) See įor instance, this kind of "fight" also happens with evaporation inside a closed recipient. The speed of bonds breaking and the speed of recombination "fight" one another, until they are in chemical equilibrium, that is when both speeds are the same. Note that even though H+ and OH- are naturally produced in water, they also recombine back into H2O. By the way, that is what makes both pH and pOH of water equal 7. that is: covalent bonds are breaking all the time (self-ionization), just like intermolecular bonds (evaporation). 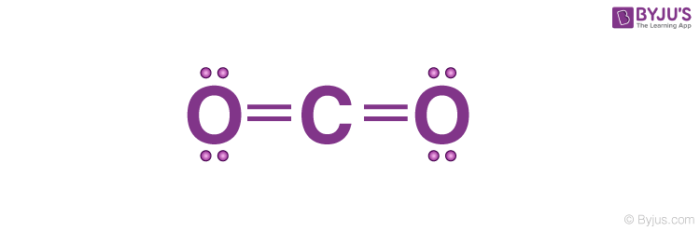
The concentration of each of these ions in pure water, at 25☌, and pressure of 1atm, is 1.0×10e−7mol/L. But, then, why no hydrogen or oxygen is observed as a product of pure water? Because water decomposes into H+ and OH- when the covalent bond breaks. Water, for example is always evaporating, even if not boiling. Yes, they can both break at the same time, it is just a matter of probability. Statistically, intermolecular bonds will break more often than covalent or ionic bonds. Intermolecular bonds break easier, but that does not mean first.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
